鱼IL-6ELISA试剂盒说明书
产品属性
产品评论
相关试剂
免费代测
常见问题
鱼IL-6ELISA试剂盒说明书
产品属性
产品评论
相关试剂
免费代测
常见问题
鱼IL-6ELISA试剂盒试验原理:
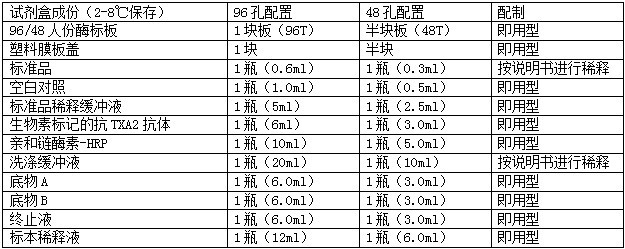
鱼IL-6ELISA试剂盒是固相夹心法酶联免疫吸附实验(ELISA).已知待测物质浓度的标准品、未知浓度的样品加入微孔酶标板内进行检测。先将待测物质和生物素标记的抗体同时温育。洗涤后,加入亲和素标记过的HRP。再经过温育和洗涤,去除未结合的酶结合物,然后加入底物A、B,和酶结合物同时作用。产生颜色。颜色的深浅和样品中待测物质的浓度呈比例关系。鱼IL-6ELISA试剂盒试剂盒内容及其配制
鱼IL-6ELISA试剂盒自备材料
1)蒸馏水。
2)加样器:5ul、10ul、50ul、100ul、200ul、500ul、1000ul。
3)振荡器及磁力搅拌器等。鱼IL-6ELISA试剂盒安全性
1)避免直接接触终止液和底物A、B。一旦接触到这些液体,请尽快用水冲洗。
2)实验中不要吃喝、抽烟或使用化妆品。
3)不要用嘴吸取试剂盒里的任何成份。鱼IL-6ELISA试剂盒操作注意事项
1)试剂应按标签说明书储存,使用前恢复到室温。稀稀过后的标准品应丢弃,不可保存。
2)实验中不用的板条应立即放回包装袋中,密封保存,以免变质。
3)不用的其它试剂应包装好或盖好。不同批号的试剂不要混用。保质前使用。
4)使用一次性的吸头以免交叉污染,吸取终止液和底物A、B液时,避免使用带金属部分的加样器。
5)使用干净的塑料容器配置洗涤液。使用前充分混匀试剂盒里的各种成份及样品。
6)洗涤酶标板时应充分拍干,不要将吸水纸直接放入酶标反应孔中吸水。
7)底物A应挥发,避免长时间打开盖子。底物B对光敏感,避免长时间暴露于光下。避免用手接触,有毒。实验完成后应立即读取OD值。
8)加入试剂的顺序应一致,以保证所有反应板孔温育的时间一样。
9)按照说明书中标明的时间、加液的量及顺序进行温育操作。鱼IL-6ELISA试剂盒样品收集、处理及保存方法
1)血清-----操作过程中避免任何细胞刺激。使用不含热原和内毒素的试管。收集血液后,1000×g离心10分钟将血清和红细胞迅速小心地分离。
2)血浆-----EDTA、柠檬酸盐、肝素血浆可用于检测。1000×g离心30分钟去除颗粒。
3)细胞上清液---1000×g离心10分钟去除颗粒和聚合物。
4)组织匀浆-----将组织加入适量生理盐水捣碎。1000×g离心10分钟,取上清液
5)保存------如果样品不立即使用,应将其分成小部分-70 ℃保存,避免反复冷冻。尽可能的不要使用溶血或高血脂血。如果血清中大量颗粒,检测前先离心或过滤。不要在37℃或更高的温度加热解冻。应在室温下解冻并确保样品均匀地充分解冻。鱼IL-6ELISA试剂盒试剂的准备
1)标准品:标准品的系列稀释应在实验时准备,不能储存。稀释前将标准品振荡混匀。
2)洗涤缓冲液(50×)的稀释:蒸馏水50倍稀释。鱼IL-6ELISA试剂盒操作步骤
1)使用前,将所有试剂充分混匀。不要使液体产生大量的泡沫,以免加样时加入大量的气泡,产生加样上的误差。
2)根据待测样品数量加上标准品的数量决定所需的板条数。每个标准品和空白孔建议做复孔。每个样品根据自己的数量来定,能使用复孔的尽量做复孔。标本用标本稀释液1:1稀释后加入50ul于反应孔内。
3)加入稀释好后的标准品50ul于反应孔、加入待测样品50ul于反应孔内。立即加入50ul的生物素标记的抗体。盖上膜板,轻轻振荡混匀,37℃温育1小时。
4)甩去孔内液体,每孔加满洗涤液,振荡30秒,甩去洗涤液,用吸水纸拍干。重复此操作3次。如果用洗板机洗涤,洗涤次数增加一次。
5)每孔加入80ul的亲和链酶素-HRP,轻轻振荡混匀,37℃温育30分钟。
6)甩去孔内液体,每孔加满洗涤液,振荡30秒,甩去洗涤液,用吸水纸拍干。重复此操作3次。如果用洗板机洗涤,洗涤次数增加一次。
7)每孔加入底物A、B各50ul,轻轻振荡混匀,37℃温育10分钟。避免光照。
8)取出酶标板,迅速加入50ul终止液,加入终止液后应立即测定结果。
9)在450nm波长处测定各孔的OD值。鱼IL-6ELISA试剂盒局限
6号标准品以上的结果为非线性的,根据此标准曲线无法得到精确的结果。鱼IL-6ELISA试剂盒试剂盒性能
1. 灵敏度:最小的检测浓度小于1号标准品。稀释度的线性。样品线性回归与预期浓度相关系数R值为0.990。
2. 特异性:不与其它细胞因子反应。
3. 重复性:板内、板间变异系数均小于10%。鱼IL-6ELISA试剂盒结果判断与分析
1、仪器值:于波长450nm的酶标仪上读取各孔的OD值
2、以吸光度OD值为纵坐标(Y),相应的待测物质标准品浓度为横坐标(X),做得相应的曲线,样品的待测物质含量可根据其OD值由标准曲线换算出相应的浓度。
产品标签
用户评论(共6条评论)
- 犬白介素18(IL18)ELISA检测试剂盒
- 犬中性粒细胞弹性蛋白酶(ELA2)ELISA检测试剂盒
- 犬载脂蛋白C1(APOC1)ELISA检测试剂盒
- 犬白介素5(IL5)ELISA检测试剂盒
- 犬白介素4(IL4)ELISA检测试剂盒
- 犬肌型肌酸激酶(CKM)ELISA检测试剂盒
- 犬组织蛋白酶K(CTSK)ELISA检测试剂盒
- 犬半乳糖凝集素9(GAL9)ELISA检测试剂盒
- 犬基质金属蛋白酶抑制因子2(TIMP2)ELISA检测试剂盒
- 犬白介素3(IL3)ELISA检测试剂盒
- 犬CD6分子(CD6)ELISA检测试剂盒
- 犬肾上腺髓质素(ADM)ELISA检测试剂盒
- 犬巨噬细胞炎性蛋白3α(MIP3α)ELISA检测试剂盒
- 犬E选择素(SELE)ELISA检测试剂盒
- 犬白介素17(IL17)ELISA检测试剂盒
- 犬单核细胞趋化蛋白4(MCP4)ELISA检测试剂盒
- 犬单核细胞趋化蛋白2(MCP2)ELISA检测试剂盒
- 犬肾素(REN)ELISA检测试剂盒
- 犬二氢硫辛酸转乙酰基酶(DLAT)ELISA检测试剂盒
- 犬红细胞生成素(EPO)ELISA检测试剂盒
- 犬载脂蛋白H(APOH)ELISA检测试剂盒
- 犬白介素1受体拮抗剂(IL1RA)ELISA检测试剂盒
- 犬干扰素γ(IFNγ)ELISA检测试剂盒
- 犬补体3a(C3a)ELISA检测试剂盒
- 犬血栓素B2(TXB2)ELISA检测试剂盒
- 犬促微管蛋白聚合蛋白(TPPP)ELISA检测试剂盒
- 犬胰岛素(INS)ELISA检测试剂盒
- 犬肌钙蛋白I(TnI)ELISA检测试剂盒
- 犬正常T细胞表达和分泌因子(RANTES)ELISA检测试剂盒
- 犬白血病抑制因子(LIF)ELISA检测试剂盒
- 犬CD40配体(CD40L)ELISA检测试剂盒
- 犬白介素8(IL8)ELISA检测试剂盒
- 犬补体片段3a受体1(C3aR1)ELISA检测试剂盒
- 犬促甲状腺素(TSH)ELISA检测试剂盒
- 犬血栓调节蛋白(TM)ELISA检测试剂盒
- 犬前列腺酸性磷酸酶(ACP3)ELISA检测试剂盒
- 犬白介素2(IL2)ELISA检测试剂盒
- 犬干细胞因子(SCF)ELISA检测试剂盒
- 犬间隙连接蛋白43(CX43)ELISA检测试剂盒
- 犬血小板生成素(TPO)ELISA检测试剂盒
- 犬粒细胞巨噬细胞集落刺激因子(GMCSF)ELISA检测试剂盒
- 犬胆囊收缩素A受体(CCKAR)ELISA检测试剂盒
- 犬胃泌素(GT)ELISA检测试剂盒
- 犬巨噬细胞炎性蛋白1β(MIP1β)ELISA检测试剂盒
- 犬多巴胺受体D2(DRD2)ELISA检测试剂盒
- 犬肌红蛋白(MYO)ELISA检测试剂盒
- 犬窖蛋白(Cav1)ELISA检测试剂盒
- 犬丙酮酸激酶(PK)ELISA检测试剂盒
- 犬心钠肽(ANP)ELISA检测试剂盒
- 犬巨噬细胞炎性蛋白1α(MIP1α)ELISA检测试剂盒
- 犬泛素(Ub)ELISA检测试剂盒
- 犬促肾上腺皮质激素(ACTH)ELISA检测试剂盒
- 犬肝素辅因子Ⅱ(HCⅡ)ELISA检测试剂盒
- 犬内皮型一氧化氮合成酶(NOS3)ELISA检测试剂盒
- 犬补体因子B(CFB)ELISA检测试剂盒
- 犬转移生长因子β1(TGFβ1)ELISA检测试剂盒
- 犬免疫球蛋白G(IgG)ELISA检测试剂盒
- 犬基质金属蛋白酶9(MMP9)ELISA检测试剂盒
- 犬甲胎蛋白(αFP)ELISA检测试剂盒
- 犬C肽(C-Peptide)ELISA检测试剂盒
- 品质保证
防伪易碎商标贴,四招助您分辨真假。 - 送货保障
产品均有现货,多家快递公司合作准时到达。 - 售后服务
产品及实验问题,提供技术支持保障。
上海酶联生物科技有限公司
Shanghai Enzyme-linked Biotechnology Co., Ltd.
订购热线:4008-898-798 / 021-54720761
传 真:021-54222852
地址:上海市松江区云凯路66号T2楼1603室
 酶联官方手机二维码
酶联官方手机二维码
微信扫描二维码,参与酶联活动,
劲爆优惠触手可得!

 © 2008-2023 沪ICP备12045995号-7 上海酶联生物科技有限公司保留所有权利
© 2008-2023 沪ICP备12045995号-7 上海酶联生物科技有限公司保留所有权利
上海酶联生物科技有限公司(www.mlbio.cn)专业提供:elisa试剂盒,ELISA试剂盒批发,进口Elisa试剂盒,酶联免疫试剂盒
本网站销售的所有产品仅用于工业应用或者科学研究等非医疗目的,不可用于人类或动物的临床诊断或治疗,非药用,非食用。